Hydrogen Cell Car
Hydrogen Cells
Hydrogen fuel cells converts water or sources of high hydrogen into electrical power. There are many types of hydrogen fuel cells for including, Alkaline, Polymer Electrolyte, and Direct method. A fuel cell is comprised of multiple components, and each fuel cell contains an anode and a cathode. Overall the reaction should produce water, or H₂O, but to generate electricity, the fuel cell needs a hydrogen fuel source. We used distilled water in the activity, and oxygen was then mixed with the distilled water and generated power in the polymer electrolyte. To charge the fuel cell and create the initial reactions we used solar panels and high intensity lights.
Solar Panels
Solar panels collect light energy from the sun or other sources, like our lights, and converts the particles into electricity. Solar panels are composed of solar cells, which use a large area diodes. When the solar cells are exposed to sunlight, the diodes absorb photon particles and convert the sunlight into electrical energy. The energy generated from photons hitting the surface of the solar cells allows electrons to be pulled in a current, from which metal contacts in the solar cell can generate electricity. The more solar cells in a solar panel will lead to more output energy, and the quality of the cells will also generate a higher output.
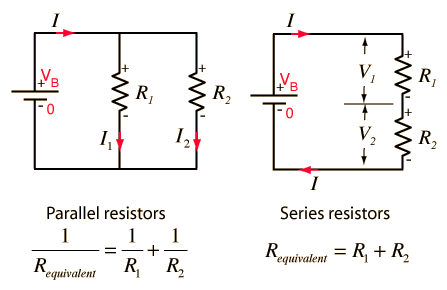
Series and Parallel Circuits
Series and parallel circuits are two of the simple forms of electrical circuits. To function the system requires a source of energy and a resistor. A series circuit contains only one path for the current to flow through. If the path is open at any given point, then the system will not work. A series circuit will have one current throughout, but voltage is split between resistors. A parallel circuit has two or more paths for the current or energy to flow through, so that if one path is unavailable then another path can be taken. Voltage will be equal across each component of the parallel circuit, but the total of the current values through each path is equal to the total current that flows from the source.
Reflection
This lab showed us how hydrogen fuel cells can be applied to the field of engineering, and it strengthened our understanding of how electricity is generated in the fuel cell. The activity gave us hands on time with the material we had been learning and gave us a break from notes. However, the instructions written in the packet can be confusing, so many groups had trouble understanding what to do at some points. I feel like a lot of time was wasted in class waiting for the fuel cells to charge up, and we would be limited in time when they were ready. On some days, people had been absent due to field trips, which allowed for groups to get help more often than days when everyone was there. Overall this activity gave insight on the workings of the hydrogen fuel cell and its efficiency compared to solar panels or batteries.
Activity 1.3.1
Fuel Cell Without Car
V= 1.5V
I= 115mA
P= .1725W
Solar Panel
V= 1.2V
I= 135mA
P= .162W
Fuel Cell With Car
V=.028V
I= 54mA
P= .00151W
Batteries (series)
V= 5.1V
I= 98.6mA
P= .502W
Batteries (parallel)
V= 5.2V
I= 118.2mA
P= .614W
V= 1.5V
I= 115mA
P= .1725W
Solar Panel
V= 1.2V
I= 135mA
P= .162W
Fuel Cell With Car
V=.028V
I= 54mA
P= .00151W
Batteries (series)
V= 5.1V
I= 98.6mA
P= .502W
Batteries (parallel)
V= 5.2V
I= 118.2mA
P= .614W
Conclusion Questions
1. Based on our findings and speeds of the vehicles, I found that the parallel system of batteries gave the most power to the motor to turn the wheels more.
2. I believe that power is the best measure because voltage and current together give the reading for power, and finding the power will be how much energy the motor is receiving to turn the wheel.
3. To achieve the same power as batteries using solar panels, we must use about 4-5 panels and wire them so that all the wires will be attached to a panel and run down to the breadboard on to the positive strip, and the energy will transfer out through wires on the negative strip going back to the solar panels.
4. To achieve the same power as batteries using fuel cells, we must use about 350-400 fuel cells and there is practical way of wiring the system without using a mega sized breadboard or multiple breadboards.
5. This hydrogen system would run overnight or during the day at work to slowly charge a battery that will run your car, and the system will run on water that the driver has to refill after after driving the car so it can recharge for the next ride.
6. Photovoltaic cells directly convert light into electricity, and some materials exhibit the photoelectric effect that causes them to absorb photons particles and release electrons out of their orbits. When these free electrons are captured, an electric current results that can be used as electricity.
Source: http://science.nasa.gov/science-news/science-at-nasa/2002/solarcells/
7. When any amount of hydrogen enters the fuel cell stack, it reacts electrochemically with oxygen to produce electric current, heat and water.
Source: http://www.fuelcellenergy.com/why-fuelcell-energy/how-do-fuel-cells-work/
2. I believe that power is the best measure because voltage and current together give the reading for power, and finding the power will be how much energy the motor is receiving to turn the wheel.
3. To achieve the same power as batteries using solar panels, we must use about 4-5 panels and wire them so that all the wires will be attached to a panel and run down to the breadboard on to the positive strip, and the energy will transfer out through wires on the negative strip going back to the solar panels.
4. To achieve the same power as batteries using fuel cells, we must use about 350-400 fuel cells and there is practical way of wiring the system without using a mega sized breadboard or multiple breadboards.
5. This hydrogen system would run overnight or during the day at work to slowly charge a battery that will run your car, and the system will run on water that the driver has to refill after after driving the car so it can recharge for the next ride.
6. Photovoltaic cells directly convert light into electricity, and some materials exhibit the photoelectric effect that causes them to absorb photons particles and release electrons out of their orbits. When these free electrons are captured, an electric current results that can be used as electricity.
Source: http://science.nasa.gov/science-news/science-at-nasa/2002/solarcells/
7. When any amount of hydrogen enters the fuel cell stack, it reacts electrochemically with oxygen to produce electric current, heat and water.
Source: http://www.fuelcellenergy.com/why-fuelcell-energy/how-do-fuel-cells-work/